Liver involvement in hyperthyroidism is uncommon in children and was first described in 1874.1 In a recent study on 204 Chinese children with hyperthyroidism, 50 were found to have abnormal liver functions.2 However, no histological biopsies or liver disease investigations were conducted in that study.
We describe here the clinical features and liver investigations including the histological features of a child who was successfully treated with carbimazole and propranolol.
Case Report
A five-year-old girl presented with a history of fever and jaundice for one week and intermittent diarrhea for a few months. She was also reported to have lost weight despite having a good appetite. Her parents denied any history of rashes, joint pains, contact with animals, or family history of chronic disorders. She was initially seen in a local hospital where the blood culture was negative, and paracetamol was prescribed. Informed written consent was obtained from the patient’s father.
Clinically, the child looked icteric, had proptosis, with a heart rate of 140/min at rest and blood pressure of 127/70 mmHg. She had wet palms and a palpable goitre with no lymphadenopathy. The liver was palpable by 4 cm in the right lobe and 7 cm in the left lobe. The spleen was not palpable. Multiple branding marks were noted over the abdominal wall. The baseline blood investigation results are listed in Table 1.
Table 1: Initial blood investigations.
|
Hb, g/dL
|
11.5–15.5
|
11.8
|
|
Albumin , g/L
|
57–82
|
20
|
|
ALT, IU/L
|
10–49
|
1676
|
|
Bilirubin, umol/L
|
5–21
|
325
|
|
GGT, IU/L
|
10–30
|
109
|
|
INR
|
0.82–1.05
|
1.4
|
|
Serum creatinine, umol/L
|
35–65
|
19
|
|
TSH, m[iU]/L
|
0.67–4
|
< 0.008
|
|
Free T4, pmol/L
|
11–18
|
83.1
|
|
Free T3, pmol/L
|
5–7
|
24.3
|
|
Thyroid peroxidase antibodies, iU/mL
|
< 60
|
177.1
|
|
Hepatitis B surface antigen
|
-
|
Negative
|
|
Alpha–fetoprotein, ug/L
|
0–20
|
13.5
|
|
Ammonia, umol/L
|
11–32
|
38
|
|
Hepatitis A, B
|
-
|
Negative
|
|
CMV IgM
|
-
|
Negative
|
|
Anti LKM antibodies
|
-
|
Negative
|
|
Autoantibodies
|
-
|
Negative
|
|
ANA, Ds-DNA; ANCA
|
-
|
Negative
|
|
IgG, g/L
|
4–12
|
22.9
|
|
IgG4, g/L
|
0.01–0.540
|
1.35
|
|
Ceruloplasmin, g/L
|
0.2–0.6
|
0.54
|
|
Neonatal screen
|
-
|
Normal
|
Hb: hemoglobulin; ALT: alanine amino transferase; GGT: gamma glutamyl transferase; INR: international normalized ratio; TSH: thyroid stimulating hormone; T: thyroid hormone; CMV: cytomegalovirus; IM: Immunoglobulin; ANA: antinuclear antibody; Ds-DNA: double stranded deoxyribonucleic acid; ANCA: antineutrophil cytoplasmic autoantibodies.
Ultrasound of the thyroid gland confirmed thyroid enlargement, the right lobe measuring 1.8 × 1.6 × 3.4 cm and the left lobe masuring 2 × 1.5 × 3.5 cm, with heterogeneous echogenicity.
Ultrasound of the liver showed coarse echotexture and patent portal and hepatic veins. Liver biopsy showed diffuse steatosis both macro and microvascular. There was a marked expansion of the portal areas along with moderate infiltration of lymphocytes, histiocytes, eosinophils, neutrophils, and occasional plasma cells. There was moderate interface inflammation in all the portal tracts. Lobular inflammation with neuroinflammatory foci was noted along with apoptotic hepatocytes, giant cell transformation, and ballooning of the hepatocyte. Also mid-focal bile duct injury was seen [Figures 1 and 2].
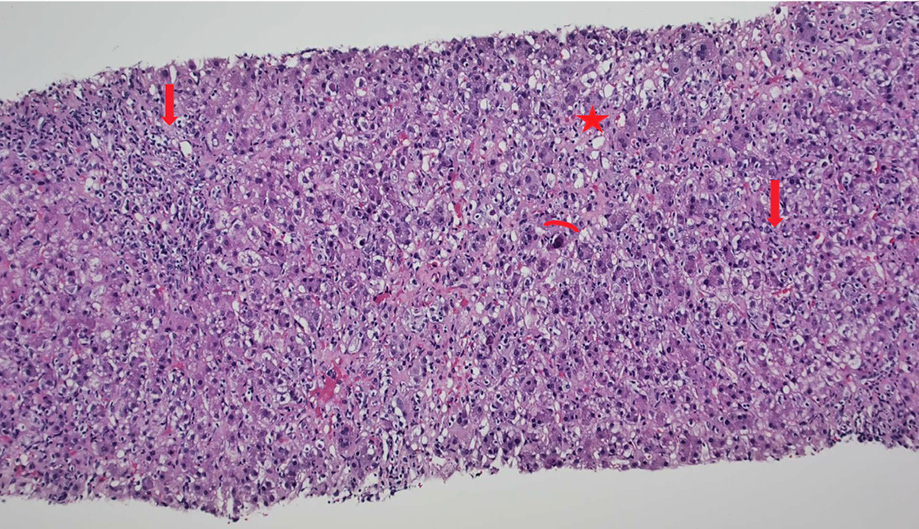 Figure 1: Sections of the liver showing disarray of the architecture with the portal and lobular inflammation (arrows), apoptosis (arch), and steatosis (star) (hematoxylin and eosin staining, magnification = 10 ×).
Figure 1: Sections of the liver showing disarray of the architecture with the portal and lobular inflammation (arrows), apoptosis (arch), and steatosis (star) (hematoxylin and eosin staining, magnification = 10 ×).
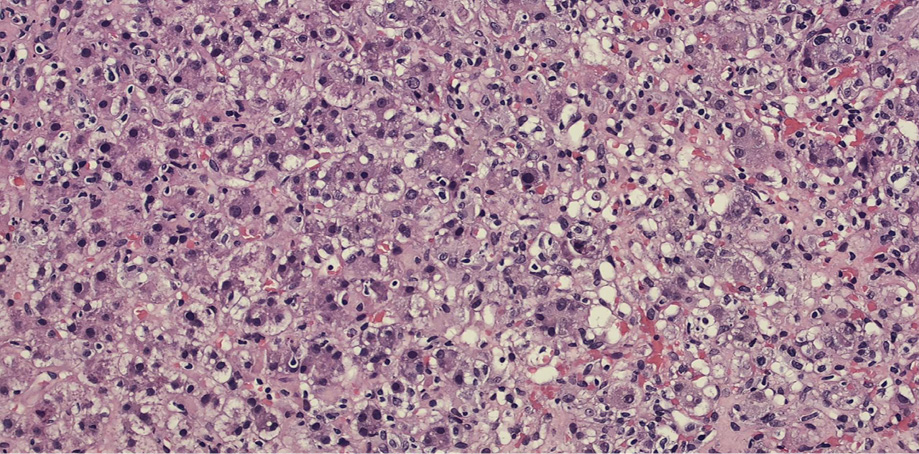 Figure 2: Sections of the liver showing steatosis with lobular disarray (hematoxylin and eosin staining, magnification = 40 ×).
Figure 2: Sections of the liver showing steatosis with lobular disarray (hematoxylin and eosin staining, magnification = 40 ×).
The histopathology was not conclusive of viral hepatitis, urea cycle defect, cholesterol ester defect, or the relation to thyrotoxicosis. In general, the child was well and had no hypoglycemia.
The patient was treated with carbimazole 5 mg three times a day (TID) (0.9 mg/kg/day), propranolol with a gradual increase to a final dose of 10 mg TID in addition to vitamin D supplements for low vitamin D of 48.6 nmol/L (50–250 nmol/L). Table 2 shows the response of liver functions to carbimazole.
Table 2: The response of ALT and bilirubin to carbimazole therapy alone.
|
Bilirubin, umol/L
|
325
|
304
|
84
|
34
|
18
|
|
ALT, IU/L
|
1676
|
208
|
57
|
26
|
32
|
|
TSH, m[iU]/L
|
< 0.008
|
|
< 0.008
|
|
< 0.008
|
ALT: alanine amino transferase; TSH: thyroid stimulating hormone; T4: thyroid hormone.
Informed written consent was obtained from the patient’s father.
Discussion
The pathophysiology of hyperthyroidism-related liver disease is not yet clear. Factors including ischemia of the hepatocytes secondary to a hypermetabolic state and relative oxygen deficiency, decreased glucuronyl transferase activity, and cardiac failure have all been proposed as potential causes.3,4 The biochemical and histopathological changes in hyperthyroidism-related liver disease have been commonly described as a mild derangement in the alanine amino transferase with mild lobular inflammation, and rarely as liver failure.5
A liver biopsy is of great value in distinguishing the likely etiology of liver disease. However, the differential diagnosis of any child presenting with autoimmune thyroid disease and liver disease must include autoimmune hepatitis. In our patient, however, histology revealed not a purely autoimmune liver disease, but with the presence of steatohepatitis with moderate necroinflammatory changes, apoptosis, and interface hepatitis. This raised a diagnostic challenge between autoimmune hepatitis and hyperthyroidism-related liver disease. The confusion between autoimmune hepatitis and thyrotoxicosis-related hepatitis was resolved by combining the clinical picture of florid thyrotoxicosis, the absence of autoantibodies against hepatocytes, the histological feature of steatohepatitis, and the presence of apoptotic hepatocytes, which were not typical features of autoimmune liver disease.
Therefore, the management of hyperthyroidism was prioritized. The rapid response to carbimazole within two weeks reassured the physicians of the primary diagnosis of thyrotoxicosis-related liver disease. Propranolol was used to keep the heart rate in a near-normal range of 80–90 beats per minute.
Urea cycle defects and cholesterol ester deficiency suggested by histopathology were not favored due to the patient’s age, absence of acidosis or neurological symptoms, and marked hyperbilirubinemia.
The first line of treatment of thyrotoxicosis in children is with an anti-thyroid drug, usually carbimazole.6 Our patient responded well to carbimazole and did not have side effects such as hepatitis, arthralgia, or agranulocytosis.
Conclusion
Hyperthyroidism-related liver disease is challenging and uncommon for children. The current case of pediatric thyrotoxicosis associated with hepatitis was diagnosed by clinical signs, liver biochemistry, biopsy, and imaging. The thyroid abnormality was corrected using carbimazole and propranolol, which also resolved the liver issues. In patients with overt hyperthyroidism and hepatitis, thorough hepatitis work-up including a liver biopsy is mandatory to clarify the exact etiology of liver disease. In patients with thyrotoxicosis, thyroid abnormalities need to be corrected first before considering autoimmune hepatitis.
Disclosure
The authors declared no conflicts of interest.
references
- 1. Habershon SO. Exophthalmic goitre, heart disease, jaundice, death. Lancet 1874;1:510-512.
- 2. Xie H, Chen D, Zhang J, Yang R, Gu W, Wang X. Characteristics of Graves’ disease in children and adolescents in Nanjing: a retrospective investigation study. Front Public Health 2022 Oct;10:993733.
- 3. Van Steenbergen W, Fevery J, De Vos R, Leyten R, Heirwegh KP, De Groote J. Thyroid hormones and the hepatic handling of bilirubin. I. Effects of hypothyroidism and hyperthyroidism on the hepatic transport of bilirubin mono- and diconjugates in the Wistar rat. Hepatology 1989 Feb;9(2):314-321.
- 4. Fong TL, McHutchison JG, Reynolds TB. Hyperthyroidism and hepatic dysfunction. A case series analysis. J Clin Gastroenterol 1992 Apr;14(3):240-244.
- 5. Sousa A, Pérez-Rodríguez MT, Páramo C, Álvarez E, Rivera A. Severe acute liver failure and thyrotoxicosis: an uncommon association. Rev Esp Enferm Dig 2015 Sep;107(9):575-576.
- 6. Léger J, Carel JC. Diagnosis and management of hyperthyroidism from prenatal life to adolescence. Best Pract Res Clin Endocrinol Metab 2018 Aug;32(4):373-386.