Chronic hepatitis B virus (HBV) infection persists and has a global impact, affecting approximately 292 million individuals worldwide.1 The repercussions of HBV infection are profound, significantly contributing to the development of life-threatening conditions such as cirrhosis and hepatocellular carcinoma (HCC).2,3 In 2019 alone, chronic HBV infections caused 820 000 deaths globally, placing a substantial burden on healthcare systems.4 In Oman, precise data on the prevalence of HBV infection remain elusive.5,6 However, estimations suggest a moderate prevalence of 2–5%, highlighting the need for ongoing vigilance and surveillance.1,6
Most global HBV infections involve inactive chronic HBV infections, with individuals in this phase of infection known as inactive carriers.7,8 According to the 2017 clinical practice guidelines of the European Association for the Study of the Liver (EASL), inactive carriers are individuals who test negative for hepatitis B e-antigen (HBeAg), positive for HBe antibody (anti-HBe), and consistently exhibit normal alanine aminotransferase (ALT) levels (< 40 IU/L) and low or undetectable serum levels of HBV DNA (< 2000 IU/mL).9 However, the EASL also acknowledges a subgroup of inactive carriers with HBV DNA levels of 2000–20 000 IU/mL, normal ALT levels, and minimal hepatic necroinflammatory activity. Regular monitoring is essential for inactive HBV carriers to detect any reactivation or potential disease progression, including routine assessment of liver fibrosis status.10
Compared to other subgroups of HBV-infected patients, individuals in the inactive carrier phase generally have a reasonably favorable prognosis.7,11 Nevertheless, the clinical profile and long-term effects of inactive infection can vary widely. For instance, some patients may have inactive infections for life, while others may experience HBV reactivation, reported in 9.4–15.9% of inactive carriers with a history of previously resolved HBV infections.7,12,13 Additionally, although uncommon, the development of HCC and liver fibrosis
remains possible.7,10,14
The significant proportion of inactive HBV carriers in the population makes this demographic particularly intriguing for research purposes, especially given that such patients often do not receive prompt medical attention.10 Furthermore, research on this specific subgroup of patients is notably limited in Oman, an area characterized by an intermediate frequency of HBV infection.1,5,6 Therefore, this study sought to describe the sociodemographic and clinical features of a cohort of adult Omani inactive hepatitis B carriers presenting to a tertiary center in Muscat, Oman. This research represents the first attempt in Oman to characterize the epidemiological and clinical presentations of individuals with inactive HBV infections.
Methods
This single-center, retrospective, descriptive study was conducted at the Sultan Qaboos University Hospital (SQUH), a tertiary care hospital in Muscat, Oman, from January 2017 to December 2018. The hospital serves patients referred from primary or secondary centers nationwide. Our target population comprised all adult Omani inactive HBV carriers who visited the adult hepatology clinic at SQUH, diagnosed with inactive chronic HBV infection, and underwent follow-up during the study period.
Inclusion criteria included adult Omani patients (aged ≥ 13 years old) diagnosed with inactive HBV infections who had undergone two-dimensional (2D) shear-wave elastography. Excluded from the study were patients with missing data, pregnant women, and those with chronic liver diseases such as hepatitis C, hepatitis D, HIV infection, autoimmune hepatitis, cholesteric liver disease, alpha one antitrypsin deficiency, Wilson’s disease, or hemochromatosis.
Relevant demographic, clinical, laboratory, and radiographical data were extracted from the patients’ electronic medical records using the hospital’s healthcare information system (TrakCare, InterSystems Corp., Cambridge, Massachusetts, USA). Information regarding anthropometric parameters, comorbidities, and family and personal history of HBV-associated risk factors was collected. As per the World Health Organization’s body mass index (BMI) cut-off points, patients were classified as underweight (< 18.5 kg/m2), normal weight (18.5–24.9 kg/m2), overweight (25.0–29.9 kg/m2), or obese (≥ 30.0 kg/m2).15 Liver stiffness measurements from ultrasonography and 2D shear wave elastography imaging were used to categorize liver fibrosis status as follows: (1) F0: no fibrosis (≤ 7.1 kPa), (2) F1: minimal fibrosis (7.1–7.8 kPa), (3) F2: significant fibrosis (7.8–8.0), (4) F3: severe fibrosis (8.0–11.5 kPa), and (5) F4: cirrhosis (> 11.5 kPa).16
We utilized SPSS (IBM Corp. Released 2020. IBM SPSS Statistics for Windows, Version 27.0. Armonk, NY: IBM Corp) for data analysis purposes. Categorical variables were presented as frequencies and percentages, while continuous variables were presented as means and SDs. Data were visually presented using frequency tables, pie charts, or bar charts.
Ethical approval for the study was obtained from the Medical Research and Ethics Committee of the College of Medicine and Health Sciences at SQU. Authorization to review patients’ medical records was granted by the relevant authorities from the Directorate of Hospital Information Systems at SQUH.
Results
A total of 375 patients with inactive HBV infections presented to the clinic during the study period; 175 (46.7%) did not meet the inclusion criteria. Thus, the final analysis included 200 patients. Of these, 106 (53.0%) were male, and the mean age was 45.0 ± 9.3 years, with most (77.5%) aged between 32–50 years [Figure 1].
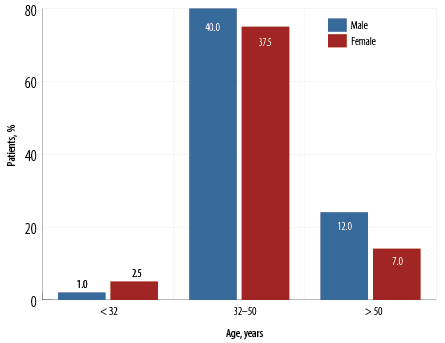 Figure 1: Age and gender distribution of adult Omani inactive hepatitis B carriers (N = 200).
Figure 1: Age and gender distribution of adult Omani inactive hepatitis B carriers (N = 200).
Patients reported a family history of chronic hepatitis B, cirrhosis, and HCC in 58 (29.0%), 22 (11.0%), and nine (4.5%) cases, respectively. Moreover, 40 (20.0%) patients had undergone surgical procedures, and five (2.5%) had received blood transfusions. No patient reported alcohol consumption. The most common comorbidities were type 2 diabetes (10.5%), dyslipidemia (9.5%), and hypertension (8.5%). Nearly half of the patients (48.5%) had BMI ≥ 25. The majority hailed from Muscat Governate, representing 38.0% of the
cohort [Table 1].
Table 1: Sociodemographic and clinical characteristics of adult Omani inactive hepatitis B carriers (N = 200).
|
Age, years
|
|
Mean ± SD
|
45.0 ± 9.3
|
|
Gender
|
|
|
Male
|
106 (53.0)
|
|
Female
|
94 (47.0)
|
|
BMI > 30
|
50 (25.0)
|
|
Relevant family history
|
|
|
Chronic HBV infection
|
58 (29.0)
|
|
Cirrhosis
|
22 (11.0)
|
|
HCC
|
9 (4.5)
|
|
Relevant medical history
|
|
|
Blood transfusions
|
5 (2.5)
|
|
Surgeries
|
40 (20.0)
|
|
Alcohol consumption
|
0 (0.0)
|
|
Comorbidities
|
|
|
T2DM
|
21 (10.5)
|
|
Dyslipidemia
|
19 (9.5)
|
|
Hypertension
|
17 (8.5)
|
|
Renal disease
|
5 (2.5)
|
|
Malignancy
|
5 (2.5)
|
|
SCD
|
3 (1.5)
|
|
Heart failure
|
1 (0.5)
|
|
Thalassemia
|
1 (0.5)
|
|
Governate destitution
|
|
|
Muscat
|
76 (38.0)
|
|
A'Dakhilyia
|
36 (18.0)
|
HBV: hepatitis B virus; HCC: hepatocellular carcinoma; T2DM: type 2 diabetes mellitus; SCD: sickle cell disease.
HBV DNA levels ranged from 2000 to 20 000 IU/mL in 33 patients (16.6%), while 132 (66.3%) had levels < 2000 IU/mL. Thirty-four patients (17.1%) had undetectable HBV DNA levels. According to 2D shear-wave elastography, most patients (80.0%) showed no (F0) to minimal fibrosis (F1); however, 32 (16.0%) and six (3.0%) exhibited stage F3 and stage F4, respectively [Table 2].
Table 2: Biochemical, viral, and radiological features of adult Omani inactive hepatitis B carriers (N = 200).
|
Liver function
|
|
|
ALT level, mean ± SD, U/L
|
22.5 ± 11.5
|
|
AST level, mean ± SD, U/L
|
19.7 ± 6.2
|
|
ALP level, mean ± SD, U/L
|
66.5 ± 19.2
|
|
Total bilirubin level, mean ± SD, μmol/L
|
9.0 ± 6.5
|
|
Albumin level, g/L, mean ± SD
|
44.9 ± 3.4
|
|
Viral load*
|
|
|
HBV DNA level, median (IQR), IU/mL
|
139.5 (20.0–779.0)
|
|
Not detected
|
34 (17.1)
|
|
0–2 000, IU/mL
|
132 (66.3)
|
|
2000–20 000, IU/mL
|
33 (16.6)
|
|
Liver fibrosis stage†
|
|
|
F0: no fibrosis
|
143 (71.5)
|
|
F1: minimal/mild fibrosis
|
17 (8.5)
|
|
F2: significant fibrosis
|
2 (1.0)
|
|
F3: severe fibrosis
|
32 (16.0)
|
ALT: alanine aminotransferase; AST: aspartate aminotransferase; ALP: alkaline phosphatase; HBV: hepatitis B virus; *Percentages for this variable are calculated from the total number of patients for whom data were available (n = 199). †As determined by two-dimentional shear-wave electrography.
Discussion
This retrospective cohort study aimed to identify the demographic and clinical characteristics of Omani patients with inactive HBV infections at a tertiary hospital in Muscat, Oman. The cohort exhibited a 1.1:1.0 gender ratio. Some studies reported a similar gender distribution among inactive HBV carriers, while others noted a male predominance, which may result from estrogen’s protective effect against chronic liver disease development.17-19 However, the intensity of male predominance in chronic HBV infection escalates with disease progression, peaking in HCC cases.19 Additionally, the reported gender differences might relate to the higher risk of bloodborne infection transmission among men due to behaviors like unprotected sex and intravenous drug use, potentially less common in highly conservative, religious countries like Oman.20
The mean age of the cohort was 45.0 years, with the majority (77.5%) born before 1990—the year Oman included the HBV vaccination in its national childhood immunization program.21 Previous reports among Omani chronic HBV patients have made similar observations.5 Notably, most female patients were of childbearing age, increasing the risk of mother-to-child transmission. Indeed, a significant portion of chronic HBV patients in Oman reports a positive family history, with 8.8% noting a maternal link.6 Correspondingly, nearly one-third of the study’s cohort had a positive family history of HBV infection, underscoring the role of vertical transmission and intra-familial contact in spreading HBV within the Omani population. Globally, especially in regions with moderate-to-high HBV incidence, vertical transmission remains the predominant method of HBV spread.22
In our study, 20.0% of patients had undergone surgical procedures. We did not assess the timing of these procedures relative to the diagnosis of HBV infection; therefore, we could not determine any correlations between these factors. However, a previous case-control study from Iran—which has a hepatitis B incidence comparable to Oman’s—identified a history of surgery as an independent risk factor for chronic HBV transmission.23 Similarly, a study among Bahraini patients with chronic HBV infections highlighted dental treatments, surgical procedures, and blood transfusions as significant transmission routes.24 Conversely, a systematic review found that surgical operations have a minimal impact on maternal HBV transmission in various Arab and African countries.25 Thus, assessing the exact impact of surgical treatments on HBV transmission remains difficult. However, the risk of this transmission route could be substantially reduced by implementing preoperative screening and rigorous sterilization and disinfection practices.
In our cohort, 71.5% of patients hailed from Muscat, A'Dakhiliyah, and Al Batinah governorates. Previous research showed that 81.6% of HBV patients reside in the northern parts of Oman.26 These findings make sense given that Muscat is the capital and most populous governorate. Therefore, one would expect a higher representation of patients in Muscat and its neighboring regions due to their greater population density. Moreover, more than two-thirds of the patients in this study were obese or overweight according to BMI metrics, aligning with the prevalence of these nutritional states in Oman’s general population.27 Other studies also report high rates of obesity and metabolic syndrome among inactive HBV carriers.28,29 This observation is crucial because researchers have noted that elevated BMI levels might increase the prevalence of steatohepatitis and accelerate liver fibrosis development.30
In our study, most patients either had undetectable HBV DNA levels or levels < 2000 IU/mL, consistent with findings from other studies on patients with inactive HBV infections.29 Moreover, inactive HBV carriers commonly exhibit an average ALT level of 22.5 ± 11 U/L, which can indicate liver damage and is a crucial metric for guiding follow-up and treatment decisions.14,31 Although inactive HBV infections were traditionally associated with lower rates of liver fibrosis, recent evidence suggests that some patients may indeed develop liver fibrosis. Transient elastography, a non-invasive method, has emerged as a promising tool for assessing fibrosis without needing a liver biopsy.16,32
Contrary findings exist in the literature regarding the prevalence of histological liver changes in inactive HBV carriers. Reports range from moderate liver fibrosis in 10% to up to 20% of patients showing significant fibrosis.33,34 In this study, 80.0% of patients exhibited none (F0) to minimal fibrosis (F1) according to 2D shear-wave elastography. However, 1.0% and 16.0% of patients displayed significant (F2) and severe fibrosis (F3), respectively, while 3.0% had developed cirrhosis (F4). Although the proportion of patients with significant fibrosis was notably lower compared to other studies using transient elastography (14–25%), the rate of cirrhosis aligned with previously reported rates (5–6%).28,29 This discrepancy in findings might stem from the absence of reported alcohol consumption history in this cohort or the exclusion of patients with co-infection history with hepatitis C, human immunodeficiency virus coinfection, and steatohepatitis—all of which pose a significant risk of liver fibrosis among inactive HBV carriers.29
This study illuminates the sociodemographic and clinical features of inactive HBV carriers in Oman, a previously unexplored area of research. The findings could guide future investigations into related research avenues, including causal relationships. Additionally, the results facilitate differentiation between healthy inactive carriers and those requiring additional medical attention due to significant fibrosis. However, it is essential to acknowledge certain limitations of this study, including potential missing or inaccurate data inherent in retrospective studies reliant on existing medical records. Furthermore, although the study was conducted at a single center, the sample size should be deemed representative of the Omani population given that SQUH serves as one of only four tertiary care hospitals in Oman and receives patients from across the country.
Conclusion
Our study aimed to describe the sociodemographic and clinical profile of a sample of inactive HBV carriers who presented to a tertiary hospital in Oman. It is notable that a significant proportion of patients were born before the integration of the HBV vaccine into the national immunization program in 1990. This underscores a heightened risk of vertical transmission, further supported by the presence of a family history of HBV infection, which was reported in nearly one-third of the participants. While the majority of patients showed either no or minimal fibrosis, a substantial subset (19.0%) exhibited severe fibrosis or cirrhosis, highlighting the diversity of outcomes in inactive carriers. Therefore, additional preventive measures are crucial, including targeted screening of at-risk age groups and the development of strategies to prevent mother-to-child transmission. These interventions are essential for improving disease management outcomes and reducing the overall disease burden associated with HBV infection in Oman.
Disclosure
The authors declared no conflicts of interest. No funding was received for this study.
references
- 1. Polaris Observatory Collaborators. Global prevalence, treatment, and prevention of hepatitis B virus infection in 2016: a modelling study. Lancet Gastroenterol Hepatol 2018 Jun;3(6):383-403.
- 2. Devarbhavi H, Asrani SK, Arab JP, Nartey YA, Pose E, Kamath PS. Global burden of liver disease: 2023 update. J Hepatol 2023 Aug;79(2):516-537.
- 3. McMahon BJ. The natural history of chronic hepatitis B virus infection. Hepatology 2009 May;49(5)(Suppl):S45-S55.
- 4. World Health Organization. Hepatitis B. 2024 [cited 2023 November]. Available from: www.who.int/news-room/fact-sheets/detail/hepatitis-b.
- 5. Al-Naamani K, Al-Maqbali A, Al-Sinani S. Characteristics of hepatitis B infection in a sample of Omani patients. Sultan Qaboos Univ Med J 2013 Aug;13(3):380-385.
- 6. Al-Busafi SA, Al-Harthi R, Al-Naamani K, Al-Zuhaibi H, Priest P. Risk factors for hepatitis B virus transmission in Oman. Oman Med J 2021 Jul;36(4):e287.
- 7. Sharma SK, Saini N, Chwla Y. Hepatitis B virus: inactive carriers. Virol J 2005 Sep;2:82.
- 8. Higgins PA. Hepatitis B virus. JAAPA 2016 Aug;29(8):48-49.
- 9. European Association for the Study of the Liver. Electronic address: [email protected]; European Association for the Study of the Liver. EASL 2017 clinical practice guidelines on the management of hepatitis B virus infection. J Hepatol 2017 Aug;67(2):370-398.
- 10. Pita I, Horta-Vale AM, Cardoso H, Macedo G. Hepatitis B inactive carriers: an overlooked population? GE Port J Gastroenterol 2014;21(6):241-249.
- 11. Ikeda K, Arase Y, Saitoh S, Kobayashi M, Someya T, Hosaka T, et al. Long-term outcome of HBV carriers with negative HBe antigen and normal aminotransferase. Am J Med 2006 Nov;119(11):977-985.
- 12. Elkady A, Aboulfotuh S, Ali EM, Sayed D, Abdel-Aziz NM, Ali AM, et al. Incidence and characteristics of HBV reactivation in hematological malignant patients in south Egypt. World J Gastroenterol 2013 Oct;19(37):6214-6220.
- 13. Chu CM, Liaw YF. Incidence and risk factors of progression to cirrhosis in inactive carriers of hepatitis B virus. Am J Gastroenterol 2009 Jul;104(7):1693-1699.
- 14. Buti M, Riveiro-Barciela M, Esteban R. Rethinking the inactive carrier state: management of patients with low-replicative HBeAg-negative chronic hepatitis B and normal liver enzymes. Clinical Dilemmas in Viral Liver Disease 2020:150-155.
- 15. World Health Organization. A healthy lifestyle- WHO recommendations. 2010 [cited 2023 December]. Available from: www.who.int/europe/news-room/fact-sheets/item/a-healthy-lifestyle---who-recommendations.
- 16. Sporea I, Bota S, Gradinaru-Taşcău O, Sirli R, Popescu A, Jurchiş A. Which are the cut-off values of 2D-shear wave elastography (2D-SWE) liver stiffness measurements predicting different stages of liver fibrosis, considering transient elastography (TE) as the reference method? Eur J Radiol 2014 Mar;83(3):e118-e122.
- 17. Tong MJ, Trieu J. Hepatitis B inactive carriers: clinical course and outcomes. J Dig Dis 2013 Jun;14(6):311-317.
- 18. Somé EN, Guingané AN, Zongo I, Ouedraogo ML, Drabo M, Sombié R. Epidemiological and clinical characteristics of inactive carriers of hepatitis B virus: a descriptive cross-sectional study in Burkina Faso. Infect Epidemiol Microbiol 2021;7(3):237-244.
- 19. Wang SH, Chen PJ, Yeh SH. Gender disparity in chronic hepatitis B: mechanisms of sex hormones. J Gastroenterol Hepatol 2015 Aug;30(8):1237-1245.
- 20. Brown R, Goulder P, Matthews PC. Sexual dimorphism in chronic hepatitis B virus (HBV) infection: evidence to inform elimination efforts. Wellcome Open Res 2022 Apr;7:32.
- 21. World Health Organization. Assessing hepatitis B in Oman. [cited 2023 December]. Available from: www.emro.who.int/omn/oman-news/assessing-hepatitis-b-in-oman.html.
- 22. MacLachlan JH, Cowie BC. Hepatitis B virus epidemiology. Cold Spring Harb Perspect Med 2015 May;5(5):a021410.
- 23. Jahangirnezhad M, Hajiani E, Makvandi M, Jalali F. A study on risk factors of chronic hepatitis B carriers. Jundishapur J Microbiol 2011;4(4):267-272.
- 24. Janahi EM. Prevalence and risk factors of hepatitis B virus infection in Bahrain, 2000 through 2010. PLoS One 2014 Feb;9(2):e87599.
- 25. Gasim GI, Murad IA, Adam I. Hepatitis B and C virus infections among pregnant women in Arab and African countries. J Infect Dev Ctries 2013 Aug;7(8):566-578.
- 26. Al Baqlani SA, Sy BT, Ratsch BA, Al Naamani K, Al Awaidy S, Busaidy SA, et al. Molecular epidemiology and genotyping of hepatitis B virus of HBsAg-positive patients in Oman. PLoS One 2014 May;9(5):e97759.
- 27. Oman Ministry of Health, World Health Organization. National health survey of non-communicable diseases risk factors. 2017 STEPS survey report. [cited 2023 December]. Available from: https://mohcsr.gov.om/wp-content/uploads/2021/03/OMAN_NCD-Risk-Factors-Survey_2017_Report.pdf.
- 28. Mena Á, Pedreira JD, Castro Á, López S, Vázquez P, Poveda E. Metabolic syndrome association with fibrosis development in chronic hepatitis B virus inactive carriers. J Gastroenterol Hepatol 2014 Jan;29(1):173-178.
- 29. Cristina SJ, Marta CM, Mercedes GS, Almudena PM, Álvaro HM, Luis VS, et al. Characterization and evaluation of liver fibrosis grade in patients with chronic hepatitis B virus infection and normal transaminases. Clin Mol Hepatol 2018 Dec;24(4):384-391.
- 30. Oliveri F, Coco B, Ciccorossi P, Colombatto P, Romagnoli V, Cherubini B, et al. Liver stiffness in the hepatitis B virus carrier: a non-invasive marker of liver disease influenced by the pattern of transaminases. World J Gastroenterol 2008 Oct;14(40):6154-6162.
- 31. Ijaz B, Ahmad W, Javed FT, Gull S, Hassan S. Revised cutoff values of ALT and HBV DNA level can better differentiate HBeAg (-) chronic inactive HBV patients from active carriers. Virol J 2011 Feb;8:86.
- 32. Pavlov CS, Casazza G, Nikolova D, Tsochatzis E, Burroughs AK, Ivashkin VT, et al. Transient elastography for diagnosis of stages of hepatic fibrosis and cirrhosis in people with alcoholic liver disease. Cochrane Database Syst Rev 2015 Jan;1(1):CD010542.
- 33. Fateen AA, Shahin RY, Farres MN, Eldeeb MA, Amer HA. Assessment of hepatic fibrosis and necroinflammation among inactive HBsAg carriers in Egypt. Ann Hepatol 2012;11(4):464-470.
- 34. Papatheodoridis GV, Manolakopoulos S, Liaw YF, Lok A. Follow-up and indications for liver biopsy in HBeAg-negative chronic hepatitis B virus infection with persistently normal ALT: a systematic review. J Hepatol 2012 Jul;57(1):196-202.